Black Fire Clip Art Most Common Isotope of Chromium
 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Manganese | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pronunciation | | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | silvery metallic | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard atomic weight A r, std(Mn) | 54.938043(2) [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Manganese in the periodic table | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic number (Z) | 25 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group | group 7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Period | period four | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Block | d-block | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Ar] 3d5 4s2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, viii, thirteen, 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concrete properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase atSTP | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | 1519 K (1246 °C, 2275 °F) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Humid point | 2334 K (2061 °C, 3742 °F) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (nighr.t.) | seven.21 g/cm3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| when liquid (atchiliad.p.) | five.95 g/cm3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of fusion | 12.91 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Estrus of vaporization | 221 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar heat capacity | 26.32 J/(mol·K) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Vapor force per unit area
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | −3, −2, −1, 0, +1, +2 , +iii , +4 , +5, +6 , +7 (depending on the oxidation state, an acidic, bones, or amphoteric oxide) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | Pauling scale: 1.55 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ionization energies |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diminutive radius | empirical: 127 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | Low spin: 139±5 pm High spin: 161±8 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Natural occurrence | primordial | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal construction | body-centered cubic (bcc) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of sound thin rod | 5150 thousand/s (at twenty °C) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal expansion | 21.7 µm/(1000⋅Yard) (at 25 °C) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal conductivity | 7.81 W/(m⋅K) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrical resistivity | ane.44 µΩ⋅m (at 20 °C) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Magnetic ordering | paramagnetic | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar magnetic susceptibility | (α) +529.0×10−6 cmthree/mol (293 K)[2] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Young's modulus | 198 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bulk modulus | 120 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mohs hardness | six.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brinell hardness | 196 MPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Number | 7439-96-5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Discovery | Carl Wilhelm Scheele (1774) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kickoff isolation | Johann Gottlieb Gahn (1774) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Main isotopes of manganese | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Manganese is a chemical element with the symbol Mn and atomic number 25. It is a hard, breakable, silvery metallic, oftentimes found in minerals in combination with iron. Manganese is a transition metal with a multifaceted assortment of industrial alloy uses, particularly in stainless steels. Information technology improves strength, workability, and resistance to clothing. Manganese oxide is used equally an oxidising agent; equally a rubber additive; and in glass making, fertilisers, and ceramics. Manganese sulfate can be used as a fungicide.
Manganese is also an essential homo dietary element, of import in macronutrient metabolism, bone formation, and free radical defense systems. Information technology is a critical component in dozens of proteins and enzymes.[three] It is found mostly in the bones, but also the liver, kidneys, and brain.[4] In the man brain, the manganese is bound to manganese metalloproteins, almost notably glutamine synthetase in astrocytes.
Manganese was first isolated in 1774. It is familiar in the laboratory in the course of the deep violet salt potassium permanganate. It occurs at the agile sites in some enzymes.[5] Of particular interest is the use of a Mn-O cluster, the oxygen-evolving complex, in the product of oxygen by plants.
Characteristics [edit]
Concrete backdrop [edit]

Electrolytically refined manganese chips and a 1 cm3 cube
Manganese is a silvery-greyness metallic that resembles iron. Information technology is hard and very breakable, difficult to fuse, but piece of cake to oxidize.[six] Manganese metal and its common ions are paramagnetic.[7] Manganese tarnishes slowly in air and oxidizes ("rusts") like iron in water containing dissolved oxygen.
Isotopes [edit]
Naturally occurring manganese is equanimous of one stable isotope, 55Mn. Several radioisotopes take been isolated and described, ranging in atomic weight from 44 u (44Mn) to 69 u (69Mn). The nearly stable are 53Mn with a one-half-life of 3.7 million years, 54Mn with a one-half-life of 312.2 days, and 52Mn with a half-life of v.591 days. All of the remaining radioactive isotopes have half-lives of less than 3 hours, and the majority of less than 1 minute. The main decay mode in isotopes lighter than the most abundant stable isotope, 55Mn, is electron capture and the primary mode in heavier isotopes is beta disuse.[eight] Manganese besides has three meta states.[viii]
Manganese is part of the atomic number 26 group of elements, which are thought to be synthesized in large stars shortly before the supernova explosion.[9] 53Mn decays to 53Cr with a half-life of 3.7 1000000 years. Because of its relatively short half-life, 53Mn is relatively rare, produced by catholic rays touch on on iron.[10] Manganese isotopic contents are typically combined with chromium isotopic contents and have found application in isotope geology and radiometric dating. Mn–Cr isotopic ratios reinforce the evidence from 26Al and 107Pd for the early history of the Solar System. Variations in 53Cr/52Cr and Mn/Cr ratios from several meteorites suggest an initial 53Mn/55Mn ratio, which indicate that Mn–Cr isotopic composition must result from in situ disuse of 53Mn in differentiated planetary bodies. Hence, 53Mn provides boosted evidence for nucleosynthetic processes immediately earlier coalescence of the Solar System.
Oxidation states [edit]

The most mutual oxidation states of manganese are +2, +3, +iv, +half-dozen, and +7, though all oxidation states from −3 to +7 accept been observed. Mntwo+ often competes with Mg2+ in biological systems, with the ion also existence very similar in its properties to Catwo+ and Zn2+. Manganese compounds where manganese is in oxidation land +7, which are mostly restricted to the unstable oxide MniiO7, compounds of the intensely purple permanganate anion MnO4 −, and a few oxyhalides (MnO3F and MnOthreeCl), are powerful oxidizing agents.[vi] Compounds with oxidation states +5 (blueish) and +6 (green) are strong oxidizing agents and are vulnerable to disproportionation.

Aqueous solution of KMnOiv illustrating the deep purple of Mn(VII) as it occurs in permanganate
The most stable oxidation state for manganese is +ii, which has a pale pink color, and many manganese(2) compounds are known, such equally manganese(Two) sulfate (MnSO4) and manganese(II) chloride (MnCl2). This oxidation state is also seen in the mineral rhodochrosite (manganese(II) carbonate). Manganese(II) near commonly exists with a high spin, S = 5/2 ground state because of the high pairing energy for manganese(Ii). However, there are a few examples of low-spin, Due south =i/two manganese(Two).[12] At that place are no spin-immune d–d transitions in manganese(Ii), explaining why manganese(Two) compounds are typically pale to colorless.[13]
| Oxidation states of manganese[14] | |
|---|---|
| 0 | Mn 2 (CO) x |
| +1 | MnC 5 H four CH 3 (CO) 3 |
| +ii | MnCl 2 , MnCO 3 , MnO |
| +3 | MnF 3 , Mn(OAc) 3 , Mn 2 O three |
| +4 | MnO 2 |
| +five | K 3 MnO 4 |
| +6 | M ii MnO 4 |
| +7 | KMnO 4 , Mn 2 O 7 |
| Mutual oxidation states are in bold. | |
The +three oxidation state is known in compounds similar manganese(III) acetate, but these are quite powerful oxidizing agents and also decumbent to disproportionation in solution, forming manganese(II) and manganese(IV). Solid compounds of manganese(3) are characterized by its stiff purple-ruby-red color and a preference for distorted octahedral coordination resulting from the Jahn-Teller event.
The oxidation country +5 can be produced by dissolving manganese dioxide in molten sodium nitrite.[15] Manganate(VI) salts can exist produced by dissolving Mn compounds, such every bit manganese dioxide, in molten alkali while exposed to air.
Permanganate (+seven oxidation state) compounds are purple, and tin can give glass a violet color. Potassium permanganate, sodium permanganate, and barium permanganate are all potent oxidizers. Potassium permanganate, also called Condy'south crystals, is a commonly used laboratory reagent because of its oxidizing backdrop; it is used as a topical medicine (for example, in the treatment of fish diseases). Solutions of potassium permanganate were amidst the first stains and fixatives to be used in the grooming of biological cells and tissues for electron microscopy.[16]
The rare oxidation country +i exists in some organomanganese compounds such as the MnC5H4CH3(CO)three compound cited in the tabular array higher up.
History [edit]
The origin of the name manganese is complex. In ancient times, two black minerals were identified from the regions of the Magnetes (either Magnesia, located within modern Hellenic republic, or Magnesia ad Sipylum, located inside modern Turkey).[17] They were both called magnes from their place of origin, only were considered to differ in sex. The male magnes attracted fe, and was the iron ore now known equally lodestone or magnetite, and which probably gave united states of america the term magnet. The female magnes ore did not attract fe, but was used to decolorize drinking glass. This female magnes was later called magnesia, known now in modern times every bit pyrolusite or manganese dioxide.[ citation needed ] Neither this mineral nor elemental manganese is magnetic. In the 16th century, manganese dioxide was called manganesum (note the two Ns instead of ane) by glassmakers, possibly equally a corruption and chain of two words, since alchemists and glassmakers eventually had to differentiate a magnesia nigra (the black ore) from magnesia alba (a white ore, also from Magnesia, too useful in glassmaking). Michele Mercati chosen magnesia nigra manganesa, and finally the metal isolated from it became known equally manganese (German: Mangan). The name magnesia eventually was then used to refer simply to the white magnesia alba (magnesium oxide), which provided the proper noun magnesium for the complimentary element when it was isolated much after.[18]

Some of the cave paintings in Lascaux, France, use manganese-based pigments.[19]
Several colorful oxides of manganese, for example manganese dioxide, are abundant in nature and have been used as pigments since the Stone Historic period. The cave paintings in Gargas that are thirty,000 to 24,000 years old contain manganese pigments.[20]
Manganese compounds were used by Egyptian and Roman glassmakers, either to add to, or remove, color from glass.[21] Employ as "glassmakers lather" continued through the Heart Ages until mod times and is evident in 14th-century glass from Venice.[22]

Considering information technology was used in glassmaking, manganese dioxide was available for experiments past alchemists, the first chemists. Ignatius Gottfried Kaim (1770) and Johann Glauber (17th century) discovered that manganese dioxide could be converted to permanganate, a useful laboratory reagent.[23] By the mid-18th century, the Swedish pharmacist Carl Wilhelm Scheele used manganese dioxide to produce chlorine. Beginning, hydrochloric acid, or a mixture of dilute sulfuric acid and sodium chloride was made to react with manganese dioxide, and afterward muriatic acid from the Leblanc process was used and the manganese dioxide was recycled by the Weldon procedure. The production of chlorine and hypochlorite bleaching agents was a large consumer of manganese ores.
Past the mid-18th century, Carl Wilhelm Scheele used pyrolusite to produce chlorine. Scheele and others were aware that pyrolusite (now known to be manganese dioxide) independent a new element. Johan Gottlieb Gahn was the first to isolate an impure sample of manganese metal in 1774, which he did by reducing the dioxide with carbon.
The manganese content of some atomic number 26 ores used in Greece led to speculations that steel produced from that ore contains boosted manganese, making the Spartan steel exceptionally hard.[24] Effectually the beginning of the 19th century, manganese was used in steelmaking and several patents were granted. In 1816, it was documented that iron alloyed with manganese was harder simply not more brittle. In 1837, British bookish James Couper noted an clan between miners' heavy exposure to manganese and a form of Parkinson'south disease.[25] In 1912, Usa patents were granted for protecting firearms against rust and corrosion with manganese phosphate electrochemical conversion coatings, and the procedure has seen widespread use ever since.[26]
The invention of the Leclanché cell in 1866 and the subsequent improvement of batteries containing manganese dioxide every bit cathodic depolarizer increased the need for manganese dioxide. Until the evolution of batteries with nickel-cadmium and lithium, well-nigh batteries contained manganese. The zinc–carbon battery and the alkali metal battery normally use industrially produced manganese dioxide because naturally occurring manganese dioxide contains impurities. In the 20th century, manganese dioxide was widely used every bit the cathodic for commercial disposable dry out batteries of both the standard (zinc–carbon) and alkaline types.[27]
Occurrence and production [edit]
Manganese comprises almost m ppm (0.1%) of the Earth'southward crust, the 12th most arable of the crust's elements.[4] Soil contains 7–9000 ppm of manganese with an average of 440 ppm.[4] The atmosphere contains 0.01 μg/m3.[4] Manganese occurs principally as pyrolusite (MnO2), braunite, (Mn2+Mnthree+ 6)(SiO12),[28] psilomelane (Ba,H2O)iiMn5Oten , and to a lesser extent every bit rhodochrosite (MnCOthree).
| | | | | |
| Manganese ore | Psilomelane (manganese ore) | Spiegeleisen is an iron alloy with a manganese content of approximately 15% | Manganese oxide dendrites on limestone from Solnhofen, Germany – a kind of pseudofossil. Scale is in mm | Mineral rhodochrosite (manganese(2) carbonate) |

Per centum of manganese output in 2006 by countries[29]
The almost of import manganese ore is pyrolusite (MnO2). Other economically important manganese ores usually show a close spatial relation to the fe ores, such as sphalerite.[6] [30] State-based resource are large merely irregularly distributed. About 80% of the known world manganese resources are in South Africa; other of import manganese deposits are in Ukraine, Commonwealth of australia, Republic of india, China, Gabonese republic and Brazil.[29] According to 1978 estimate, the sea flooring has 500 billion tons of manganese nodules.[31] Attempts to discover economically feasible methods of harvesting manganese nodules were abased in the 1970s.[32]
In South Africa, nigh identified deposits are located virtually Hotazel in the Northern Cape Province, with a 2011 estimate of 15 billion tons. In 2011 Due south Africa produced 3.4 million tons, topping all other nations.[33]
Manganese is mainly mined in S Africa, Australia, China, Gabonese republic, Brazil, Republic of india, Kazakhstan, Ghana, Ukraine and Malaysia.[34]
For the production of ferromanganese, the manganese ore is mixed with fe ore and carbon, and so reduced either in a blast furnace or in an electrical arc furnace.[35] The resulting ferromanganese has a manganese content of thirty to 80%.[six] Pure manganese used for the production of iron-free alloys is produced by leaching manganese ore with sulfuric acid and a subsequent electrowinning process.[36]
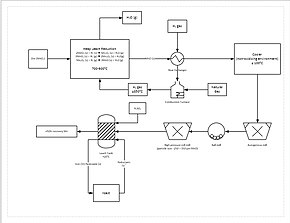
Process menstruum diagram for a manganese refining circuit.
A more than progressive extraction procedure involves directly reducing manganese ore in a heap leach. This is done by percolating natural gas through the bottom of the heap; the natural gas provides the rut (needs to be at least 850 °C) and the reducing agent (carbon monoxide). This reduces all of the manganese ore to manganese oxide (MnO), which is a leachable course. The ore then travels through a grinding circuit to reduce the particle size of the ore to between 150 and 250 μm, increasing the surface area to aid leaching. The ore is then added to a leach tank of sulfuric acid and ferrous iron (Atomic number 26ii+) in a 1.6:1 ratio. The iron reacts with the manganese dioxide to form atomic number 26 hydroxide and elemental manganese. This procedure yields approximately 92% recovery of the manganese. For further purification, the manganese tin and then exist sent to an electrowinning facility.[37]
In 1972 the CIA'southward Project Azorian, through billionaire Howard Hughes, deputed the send Hughes Glomar Explorer with the cover story of harvesting manganese nodules from the sea flooring.[38] That triggered a blitz of activeness to collect manganese nodules, which was not actually applied. The real mission of Hughes Glomar Explorer was to heighten a sunken Soviet submarine, the One thousand-129, with the goal of retrieving Soviet code books.[39]
An abundant resource of manganese in the form of Mn nodules plant on the ocean floor.[xl] [41] These nodules, which are composed of 29% manganese,[42] are located along the ocean flooring and the potential impact of mining these nodules is beingness researched. Physical, chemical, and biological ecology impacts tin can occur due to this nodule mining disturbing the seafloor and causing sediment plumes to form. This interruption includes metals and inorganic nutrients, which can lead to contagion of the almost-bottom waters from dissolved toxic compounds. Mn nodules are also the grazing grounds, living space, and protection for endo- and epifaunal systems. When theses nodules are removed, these systems are straight affected. Overall, this can cause species to leave the area or completely dice off.[43] Prior to the commencement of the mining itself, research is being conducted past United nations affiliated bodies and state-sponsored companies in an endeavor to fully understand environmental impacts in the hopes of mitigating these impacts.[44]
Oceanic environment [edit]
Many trace elements in the ocean come from metal-rich hydrothermal particles from hydrothermal vents.[45] Dissolved manganese (dMn) is institute throughout the world's oceans, 90% of which originates from hydrothermal vents.[46] Particulate Mn develops in buoyant plumes over an active vent source, while the dMn behaves conservatively.[45] Mn concentrations vary betwixt the h2o columns of the ocean. At the surface, dMn is elevated due to input from external sources such as rivers, dust, and shelf sediments. Littoral sediments normally have lower Mn concentrations, only can increase due to anthropogenic discharges from industries such as mining and steel manufacturing, which enter the ocean from river inputs. Surface dMn concentrations tin can too be elevated biologically through photosynthesis and physically from littoral upwelling and wind-driven surface currents. Internal cycling such as photograph-reduction from UV radiations tin can also drag levels by speeding up the dissolution of Mn-oxides and oxidative scavenging, preventing Mn from sinking to deeper waters.[47] Elevated levels at mid-depths can occur about mid-body of water ridges and hydrothermal vents. The hydrothermal vents release dMn enriched fluid into the water. The dMn can then travel up to four,000 km due to the microbial capsules present, preventing commutation with particles, lowing the sinking rates. Dissolved Mn concentrations are even higher when oxygen levels are low. Overall, dMn concentrations are ordinarily higher in coastal regions and decrease when moving offshore.[47]
Soils [edit]
Manganese occurs in soils in three oxidation states: the divalent cation, Mn2+ and as brownish-black oxides and hydroxides containing Mn (Three,IV), such equally MnOOH and MnO2. Soil pH and oxidation-reduction weather touch which of these three forms of Mn is ascendant in a given soil. At pH values less than 6 or under anaerobic conditions, Mn(II) dominates, while under more than alkali metal and aerobic conditions, Mn(III,Iv) oxides and hydroxides predominate. These effects of soil acerbity and aeration state on the grade of Mn can be modified or controlled by microbial activity. Microbial respiration tin cause both the oxidation of Mn2+ to the oxides, and it can cause reduction of the oxides to the divalent cation.[48]
The Mn(III,IV) oxides exist as brownish-black stains and pocket-sized nodules on sand, silt, and dirt particles. These surface coatings on other soil particles take high surface area and carry negative charge. The charged sites can adsorb and retain diverse cations, especially heavy metals (e.g., Crthree+, Cu2+, Zn2+, and Pb2+). In addition, the oxides can adsorb organic acids and other compounds. The adsorption of the metals and organic compounds can then cause them to be oxidized while the Mn(Iii,IV) oxides are reduced to Mn2+ (e.m., Crthree+ to Cr(VI) and colorless hydroquinone to tea-colored quinone polymers).[49]
Applications [edit]
Manganese has no satisfactory substitute in its major applications in metallurgy.[29] In small applications (e.g., manganese phosphating), zinc and sometimes vanadium are feasible substitutes.
Steel [edit]

Manganese is essential to atomic number 26 and steel production past virtue of its sulfur-fixing, deoxidizing, and alloying properties, as first recognized past the British metallurgist Robert Forester Mushet (1811–1891) who, in 1856, introduced the element, in the form of Spiegeleisen, into steel for the specific purpose of removing excess dissolved oxygen, sulfur, and phosphorus in guild to improve its malleability. Steelmaking,[50] including its ironmaking component, has deemed for most manganese demand, presently in the range of 85% to 90% of the total demand.[36] Manganese is a cardinal component of depression-cost stainless steel.[51] [52] Often ferromanganese (normally about eighty% manganese) is the intermediate in mod processes.
Minor amounts of manganese improve the workability of steel at high temperatures by forming a loftier-melting sulfide and preventing the germination of a liquid fe sulfide at the grain boundaries. If the manganese content reaches 4%, the embrittlement of the steel becomes a dominant feature. The embrittlement decreases at higher manganese concentrations and reaches an acceptable level at 8%. Steel containing 8 to 15% of manganese has a high tensile forcefulness of up to 863 MPa.[53] [54] Steel with 12% manganese was discovered in 1882 by Robert Hadfield and is nonetheless known every bit Hadfield steel (mangalloy). It was used for British military steel helmets and afterwards past the U.S. military.[55]
Aluminium alloys [edit]
The second largest application for manganese is in aluminium alloys. Aluminium with roughly 1.5% manganese has increased resistance to corrosion through grains that absorb impurities which would pb to galvanic corrosion.[56] The corrosion-resistant aluminium alloys 3004 and 3104 (0.8 to 1.five% manganese) are used for most potable cans.[57] Before 2000, more than than i.6 million tonnes of those alloys were used; at 1% manganese, this consumed xvi,000 tonnes of manganese.[ failed verification ] [57]
Other uses [edit]
Methylcyclopentadienyl manganese tricarbonyl is used as an additive in unleaded gasoline to heave octane rating and reduce engine knocking. The manganese in this unusual organometallic chemical compound is in the +one oxidation state.[58]
Manganese(IV) oxide (manganese dioxide, MnO2) is used as a reagent in organic chemistry for the oxidation of benzylic alcohols (where the hydroxyl grouping is next to an aromatic band). Manganese dioxide has been used since antiquity to oxidize and neutralize the greenish tinge in glass from trace amounts of iron contamination.[22] MnOii is as well used in the manufacture of oxygen and chlorine and in drying blackness paints. In some preparations, it is a dark-brown paint for paint and is a constituent of natural umber.
Tetravalent manganese is used equally an activator in red-emitting phosphors. While many compounds are known which show brilliance,[59] the bulk are not used in commercial application due to low efficiency or deep red emission.[60] [61] Notwithstanding, several Mn4+ activated fluorides were reported as potential red-emitting phosphors for warm-white LEDs.[62] [63] But to this day, but K2SiF6:Mn4+ is commercially bachelor for use in warm-white LEDs.[64]
Batteries [edit]
Manganese(IV) oxide was used in the original type of dry out jail cell battery as an electron acceptor from zinc, and is the blackish fabric in carbon–zinc type flashlight cells. The manganese dioxide is reduced to the manganese oxide-hydroxide MnO(OH) during discharging, preventing the formation of hydrogen at the anode of the battery.[65]
- MnO2 + HiiO + eastward− → MnO(OH) + OH −
The same material also functions in newer alkaline batteries (usually battery cells), which use the aforementioned basic reaction, but a different electrolyte mixture. In 2002, more than 230,000 tons of manganese dioxide was used for this purpose.[27] [65]

Globe-War-Two-era five-cent coin (1942-5 identified past mint mark P, D or S above dome) made from a 56% copper-35% silvery-9% manganese alloy
Minting [edit]
The metallic is occasionally used in coins; until 2000, the only Usa money to use manganese was the "wartime" nickel from 1942 to 1945.[66] An alloy of 75% copper and 25% nickel was traditionally used for the product of nickel coins. However, because of shortage of nickel metallic during the war, it was substituted by more than bachelor silver and manganese, thus resulting in an blend of 56% copper, 35% silver and 9% manganese. Since 2000, dollar coins, for example the Sacagawea dollar and the Presidential $1 coins, are made from a brass containing 7% of manganese with a pure copper core.[67] In both cases of nickel and dollar, the use of manganese in the money was to duplicate the electromagnetic properties of a previous identically sized and valued coin in the mechanisms of vending machines. In the case of the afterward U.Southward. dollar coins, the manganese alloy was intended to duplicate the backdrop of the copper/nickel blend used in the previous Susan B. Anthony dollar.
Ceramic coloring [edit]
Manganese compounds have been used as pigments and for the coloring of ceramics and glass. The brownish color of ceramic is sometimes the result of manganese compounds.[68] In the drinking glass manufacture, manganese compounds are used for two furnishings. Manganese(III) reacts with fe(Two) to reduce potent dark-green colour in glass by forming less-colored atomic number 26(Three) and slightly pink manganese(2), compensating for the residual color of the iron(III).[22] Larger quantities of manganese are used to produce pink colored drinking glass. In 2009, Professor Mas Subramanian and associates at Oregon State University discovered that manganese can be combined with yttrium and indium to form an intensely bluish, not-toxic, inert, fade-resistant paint, YInMn bluish, the first new bluish pigment discovered in 200 years.
Biological role [edit]
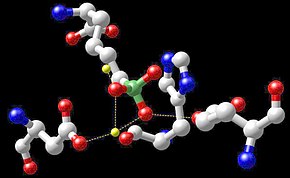
Reactive center of arginase with boronic acid inhibitor – the manganese atoms are shown in xanthous.
Deficiency [edit]
Manganese deficiency in humans results in a number of medical problems. Many common vitamin and mineral supplement products neglect to include manganese in their compositions. Relatively high dietary intake of other minerals such as iron, magnesium, and calcium may inhibit the proper intake of manganese. A deficiency of manganese causes skeletal deformation in animals and inhibits the product of collagen in wound healing. Come across Manganese deficiency
Biochemistry [edit]
The classes of enzymes that have manganese cofactors include oxidoreductases, transferases, hydrolases, lyases, isomerases and ligases. Other enzymes containing manganese are arginase and Mn-containing superoxide dismutase (Mn-SOD). Too the enzyme class of opposite transcriptases of many retroviruses (though not lentiviruses such as HIV) contains manganese. Manganese-containing polypeptides are the diphtheria toxin, lectins and integrins.[69]
Biological role in humans [edit]
Manganese is an essential human dietary element. It is present as a coenzyme in several biological processes, which include macronutrient metabolism, os formation, and free radical defence force systems. It is a critical component in dozens of proteins and enzymes.[3] The human being body contains about 12 mg of manganese, mostly in the basic. The soft tissue remainder is concentrated in the liver and kidneys.[4] In the human encephalon, the manganese is jump to manganese metalloproteins, nearly notably glutamine synthetase in astrocytes.[lxx]
Toxicity [edit]
Excessive exposure or intake may lead to a condition known as manganism, a neurodegenerative disorder that causes dopaminergic neuronal death and symptoms similar to Parkinson'due south disease.[4] [71]
Toxicity in marine life [edit]
Many enzymatic systems demand Mn to function, only in loftier levels, Mn can go toxic. One ecology reason Mn levels tin can increment in seawater is when hypoxic periods occur.[72] Since 1990 there accept been reports of Mn accumulation in marine organisms including fish, crustaceans, mollusks, and echinoderms. Specific tissues are targets in different species, including the gills, brain, blood, kidney, and liver/hepatopancreas. Physiological effects have been reported in these species. Mn can impact the renewal of immunocytes and their functionality, such equally phagocytosis and activation of pro-phenoloxidase, suppressing the organisms' immune systems. This causes the organisms to be more susceptible to infections. As climatic change occurs, pathogen distributions increment, and in lodge for organisms to survive and defend themselves against these pathogens, they need a healthy, stiff allowed system. If their systems are compromised from loftier Mn levels, they will not be able to fight off these pathogens and die.[46]
Nutrition [edit]
Dietary recommendations [edit]
| Males | Females | ||
|---|---|---|---|
| Age | AI (mg/mean solar day) | Age | AI (mg/day) |
| i–3 | one.2 | 1–three | one.2 |
| 4–8 | 1.5 | 4–8 | i.5 |
| ix–13 | i.nine | nine–13 | 1.6 |
| 14–18 | 2.2 | 14–18 | i.6 |
| xix+ | 2.3 | nineteen+ | i.8 |
| meaning: 2 | |||
| lactating: 2.6 | |||
The U.Due south. Institute of Medicine (IOM) updated Estimated Boilerplate Requirements (EARs) and Recommended Dietary Allowances (RDAs) for minerals in 2001. For manganese at that place was non sufficient information to fix EARs and RDAs, and then needs are described equally estimates for Adequate Intakes (AIs). As for safety, the IOM sets Tolerable upper intake levels (ULs) for vitamins and minerals when prove is sufficient. In the case of manganese the developed UL is gear up at xi mg/mean solar day. Collectively the EARs, RDAs, AIs and ULs are referred to equally Dietary Reference Intakes (DRIs).[73] Manganese deficiency is rare.[74]
The European Nutrient Condom Authorization (EFSA) refers to the collective set of information equally Dietary Reference Values, with Population Reference Intake (PRI) instead of RDA, and Average Requirement instead of EAR. AI and UL defined the same as in United States. For people ages xv and older the AI is set up at 3.0 mg/day. AIs for pregnancy and lactation is iii.0 mg/twenty-four hours. For children ages i–14 years the AIs increase with historic period from 0.five to 2.0 mg/twenty-four hour period. The adult AIs are college than the U.Southward. RDAs.[75] The EFSA reviewed the same safe question and decided that there was bereft information to set a UL.[76]
For U.S. nutrient and dietary supplement labeling purposes the amount in a serving is expressed as a percent of Daily Value (%DV). For manganese labeling purposes 100% of the Daily Value was 2.0 mg, only as of 27 May 2016 it was revised to two.iii mg to bring it into agreement with the RDA.[77] [78] A table of the old and new adult daily values is provided at Reference Daily Intake.
Biological role in leaner [edit]
Mn-SOD is the type of SOD nowadays in eukaryotic mitochondria, and also in most bacteria (this fact is in keeping with the bacterial-origin theory of mitochondria). The Mn-SOD enzyme is probably i of the most aboriginal, for nearly all organisms living in the presence of oxygen utilise it to deal with the toxic effects of superoxide (O −
ii ), formed from the 1-electron reduction of dioxygen. The exceptions, which are all bacteria, include Lactobacillus plantarum and related lactobacilli, which use a different nonenzymatic machinery with manganese (Mnii+) ions complexed with polyphosphate, suggesting a path of evolution for this office in aerobic life.
Biological role in plants [edit]
Manganese is also of import in photosynthetic oxygen evolution in chloroplasts in plants. The oxygen-evolving circuitous (OEC) is a part of photosystem 2 contained in the thylakoid membranes of chloroplasts; information technology is responsible for the terminal photooxidation of water during the low-cal reactions of photosynthesis, and has a metalloenzyme cadre containing four atoms of manganese.[79] [80] To fulfill this requirement, nearly broad-spectrum plant fertilizers comprise manganese.
Precautions [edit]
| Hazards | |
|---|---|
| GHS labelling: | |
| Hazard statements | H401 |
| Precautionary statements | P273, P501 [81] |
| NFPA 704 (fire diamond) | 0 0 0 |
Manganese compounds are less toxic than those of other widespread metals, such every bit nickel and copper.[82] However, exposure to manganese dusts and fumes should non exceed the ceiling value of 5 mg/thou3 even for short periods considering of its toxicity level.[83] Manganese poisoning has been linked to impaired motor skills and cognitive disorders.[84]
Permanganate exhibits a higher toxicity than manganese(II) compounds. The fatal dose is about ten g, and several fatal intoxications have occurred. The stiff oxidative outcome leads to necrosis of the mucous membrane. For example, the esophagus is affected if the permanganate is swallowed. Only a limited amount is captivated by the intestines, but this modest amount shows severe effects on the kidneys and on the liver.[85] [86]
Manganese exposure in U.s. is regulated past the Occupational Safety and Wellness Administration (OSHA).[87] People can be exposed to manganese in the workplace past breathing it in or swallowing information technology. OSHA has set the legal limit (permissible exposure limit) for manganese exposure in the workplace as 5 mg/miii over an 8-hour workday. The National Constitute for Occupational Condom and Wellness (NIOSH) has set a recommended exposure limit (REL) of i mg/mthree over an 8-hour workday and a short term limit of 3 mg/yard3. At levels of 500 mg/g3, manganese is immediately dangerous to life and wellness.[88]
Generally, exposure to ambient Mn air concentrations in backlog of 5 μg Mn/m3 can lead to Mn-induced symptoms. Increased ferroportin protein expression in human embryonic kidney (HEK293) cells is associated with decreased intracellular Mn concentration and adulterate cytotoxicity, characterized past the reversal of Mn-reduced glutamate uptake and macerated lactate dehydrogenase leakage.[89]
Ecology health concerns [edit]
In drinking h2o [edit]
Waterborne manganese has a greater bioavailability than dietary manganese. According to results from a 2010 study,[90] higher levels of exposure to manganese in drinking h2o are associated with increased intellectual impairment and reduced intelligence quotients in school-age children. Information technology is hypothesized that long-term exposure due to inhaling the naturally occurring manganese in shower water puts up to 8.7 meg Americans at risk.[91] However, data indicates that the human torso can recover from certain adverse effects of overexposure to manganese if the exposure is stopped and the body can articulate the excess.[92]
In gasoline [edit]

Methylcyclopentadienyl manganese tricarbonyl (MMT) is a gasoline additive used to replace lead compounds for unleaded gasolines to improve the octane rating of low octane petroleum distillates. It reduces engine knock agent through the activeness of the carbonyl groups. Fuels containing manganese tend to grade manganese carbides, which impairment exhaust valves. Compared to 1953, levels of manganese in air take dropped.[93]
In tobacco smoke [edit]
The tobacco plant readily absorbs and accumulates heavy metals such as manganese from the surrounding soil into its leaves. These are later on inhaled during tobacco smoking.[94] While manganese is a constituent of tobacco smoke,[95] studies have largely ended that concentrations are not chancy for human wellness.[96]
Function in neurological disorders [edit]
Manganism [edit]
Manganese overexposure is most oft associated with manganism, a rare neurological disorder associated with excessive manganese ingestion or inhalation. Historically, persons employed in the production or processing of manganese alloys[97] [98] have been at chance for developing manganism; still, current wellness and condom regulations protect workers in developed nations.[87] The disorder was first described in 1837 by British academic John Couper, who studied two patients who were m.[25]
Manganism is a biphasic disorder. In its early on stages, an intoxicated person may experience depression, mood swings, compulsive behaviors, and psychosis. Early neurological symptoms give way to late-phase manganism, which resembles Parkinson's disease. Symptoms include weakness, monotone and slowed oral communication, an expressionless face, tremor, forrard-leaning gait, inability to walk backwards without falling, rigidity, and general problems with dexterity, gait and balance.[25] [99] Unlike Parkinson'southward disease, manganism is not associated with loss of the sense of smell and patients are typically unresponsive to treatment with L-DOPA.[100] Symptoms of late-stage manganism become more severe over fourth dimension even if the source of exposure is removed and brain manganese levels render to normal.[99]
Chronic manganese exposure has been shown to produce a parkinsonism-like affliction characterized past movement abnormalities.[101] This status is not responsive to typical therapies used in the treatment of PD, suggesting an culling pathway than the typical dopaminergic loss within the substantia nigra.[101] Manganese may accumulate in the basal ganglia, leading to the abnormal movements.[102] A mutation of the SLC30A10 gene, a manganese efflux transporter necessary for decreasing intracellular Mn, has been linked with the development of this Parkinsonism-similar disease.[103] The Lewy bodies typical to PD are not seen in Mn-induced parkinsonism.[102]
Animal experiments have given the opportunity to examine the consequences of manganese overexposure nether controlled conditions. In (not-ambitious) rats, manganese induces mouse-killing behavior.[104]
Babyhood developmental disorders [edit]
Several recent studies attempt to examine the effects of chronic low-dose manganese overexposure on child development. The earliest report was conducted in the Chinese province of Shanxi. Drinking water in that location had been contaminated through improper sewage irrigation and independent 240–350 μg Mn/L. Although Mn concentrations at or below 300 μg Mn/Fifty were considered prophylactic at the fourth dimension of the study past the Us EPA and 400 μg Mn/L by the World Health Arrangement, the 92 children sampled (between 11 and xiii years of age) from this province displayed lower performance on tests of manual dexterity and rapidity, brusque-term memory, and visual identification, compared to children from an uncontaminated area. More than recently, a written report of ten-year-erstwhile children in Bangladesh showed a relationship between Mn concentration in well water and diminished IQ scores. A 3rd study conducted in Quebec examined school children between the ages of 6 and fifteen living in homes that received water from a well containing 610 μg Mn/50; controls lived in homes that received water from a 160 μg Mn/L well. Children in the experimental grouping showed increased hyperactive and oppositional behavior.[xc]
The current maximum safe concentration nether EPA rules is l μg Mn/L.[105]
Neurodegenerative diseases [edit]
A protein called DMT1 is the major transporter in manganese assimilation from the intestine, and may exist the major transporter of manganese beyond the blood–brain bulwark. DMT1 also transports inhaled manganese across the nasal epithelium. The proposed machinery for manganese toxicity is that dysregulation leads to oxidative stress, mitochondrial dysfunction, glutamate-mediated excitotoxicity, and aggregation of proteins.[106]
Encounter also [edit]
- Manganese exporter, membrane transport protein
- List of countries by manganese production
- Parkerizing
References [edit]
- ^ "Standard Atomic Weights: Manganese". CIAAW. 2017.
- ^ Weast, Robert (1984). CRC, Handbook of Chemistry and Physics. Boca Raton, Florida: Chemical Rubber Company Publishing. pp. E110. ISBN0-8493-0464-four.
- ^ a b Erikson, Keith Yard.; Ascher, Michael (2019). "Chapter 10. Manganese: Its Role in Illness and Wellness". In Sigel, Astrid; Freisinger, Eva; Sigel, Roland K. O.; Carver, Peggy L. (Invitee editor) (eds.). Essential Metals in Medicine:Therapeutic Use and Toxicity of Metallic Ions in the Clinic. Metal Ions in Life Sciences. Vol. 19. Berlin: de Gruyter GmbH. pp. 253–266. doi:x.1515/9783110527872-016. ISBN978-3-eleven-052691-2. PMID 30855111. S2CID 73725546.
- ^ a b c d e f Emsley, John (2001). "Manganese". Nature'due south Building Blocks: An A-Z Guide to the Elements. Oxford, Britain: Oxford Academy Press. pp. 249–253. ISBN978-0-19-850340-8.
- ^ Roth, Jerome; Ponzoni, Silvia; Aschner, Michael (2013). "Chapter six Manganese Homeostasis and Transport". In Banci, Lucia (ed.). Metallomics and the Cell. Metal Ions in Life Sciences. Vol. 12. Springer. pp. 169–201. doi:10.1007/978-94-007-5561-1_6. ISBN978-94-007-5560-iv. PMC6542352. PMID 23595673. Electronic-book ISBN 978-94-007-5561-1.
- ^ a b c d Holleman, Arnold F.; Wiberg, Egon; Wiberg, Nils (1985). "Mangan". Lehrbuch der Anorganischen Chemie (in German) (91–100 ed.). Walter de Gruyter. pp. 1110–1117. ISBN978-3-11-007511-three.
- ^ Lide, David R. (2004). Magnetic susceptibility of the elements and inorganic compounds, in Handbook of Chemistry and Physics. CRC press. ISBN978-0-8493-0485-9. Archived from the original on 17 December 2019. Retrieved 7 September 2019.
- ^ a b Audi, G.; Kondev, F. G.; Wang, K.; Huang, Due west. J.; Naimi, S. (2017). "The NUBASE2016 evaluation of nuclear properties" (PDF). Chinese Physics C. 41 (3): 030001. Bibcode:2017ChPhC..41c0001A. doi:10.1088/1674-1137/41/3/030001.
- ^ Clery, Daniel (4 June 2020). "The milky way's brightest explosions get nuclear with an unexpected trigger: pairs of dead stars". Science . Retrieved 26 July 2021.
{{cite web}}: CS1 maint: url-status (link) - ^ Schaefer, Jeorg; Faestermann, Thomas; Herzog, Gregory F.; Knie, Klaus; Korschinek, Gunther; Masarik, Jozef; Meier, Astrid; Poutivtsev, Michail; Rugel, Georg; Schlüchter, Christian; Serifiddin, Feride; Winckler, Gisela (2006). "Terrestrial manganese-53 – A new monitor of Earth surface processes". Earth and Planetary Scientific discipline Letters. 251 (3–4): 334–345. Bibcode:2006E&PSL.251..334S. doi:x.1016/j.epsl.2006.09.016.
- ^ "Ch. 20". Shriver and Atkins' Inorganic Chemical science. Oxford University Press. 2010. ISBN978-0-19-923617-vi.
- ^ Saha, Amrita; Majumdar, Partha; Goswami, Sreebrata (2000). "Low-spin manganese(II) and cobalt(III) complexes of Northward-aryl-ii-pyridylazophenylamines: new tridentate Due north,N,North-donors derived from cobalt mediated aromatic ring amination of 2-(phenylazo)pyridine. Crystal structure of a manganese(2) complex". Periodical of the Chemical Gild, Dalton Transactions (11): 1703–1708. doi:x.1039/a909769d.
- ^ Rayner-Canham, Geoffrey and Overton, Tina (2003) Descriptive Inorganic Chemistry, Macmillan, p. 491, ISBN 0-7167-4620-four.
- ^ Schmidt, Max (1968). "Seven. Nebengruppe". Anorganische Chemie Ii (in High german). Wissenschaftsverlag. pp. 100–109.
- ^ Temple, R. B.; Thickett, One thousand. W. (1972). "The formation of manganese(v) in molten sodium nitrite". Australian Journal of Chemistry. 25 (three): 55. doi:ten.1071/CH9720655.
- ^ Luft, J. H. (1956). "Permanganate – a new fixative for electron microscopy". Journal of Biophysical and Biochemical Cytology. 2 (vi): 799–802. doi:10.1083/jcb.2.6.799. PMC2224005. PMID 13398447.
- ^ languagehat (28 May 2005). "MAGNET". languagehat.com . Retrieved 18 June 2020.
- ^ Calvert, J. B. (24 January 2003). "Chromium and Manganese". Archived from the original on 31 December 2016. Retrieved 30 April 2009.
{{cite spider web}}: CS1 maint: bot: original URL status unknown (link) - ^
- ^ Chalmin, East.; Vignaud, C.; Salomon, H.; Farges, F.; Susini, J.; Menu, M. (2006). "Minerals discovered in paleolithic blackness pigments by manual electron microscopy and micro-X-ray absorption near-border structure" (PDF). Applied Physics A. 83 (12): 213–218. Bibcode:2006ApPhA..83..213C. doi:ten.1007/s00339-006-3510-vii. hdl:2268/67458. S2CID 9221234.
- ^ Sayre, Eastward. Five.; Smith, R. W. (1961). "Compositional Categories of Ancient Glass". Scientific discipline. 133 (3467): 1824–1826. Bibcode:1961Sci...133.1824S. doi:ten.1126/science.133.3467.1824. PMID 17818999. S2CID 25198686.
- ^ a b c Mccray, Westward. Patrick (1998). "Glassmaking in renaissance Italia: The innovation of venetian cristallo". JOM. l (5): 14–xix. Bibcode:1998JOM....50e..14M. doi:ten.1007/s11837-998-0024-0. S2CID 111314824.
- ^ Rancke-Madsen, E. (1975). "The Discovery of an Chemical element". Centaurus. 19 (4): 299–313. Bibcode:1975Cent...19..299R. doi:10.1111/j.1600-0498.1975.tb00329.10.
- ^ Alessio, L.; Campagna, K.; Lucchini, R. (2007). "From lead to manganese through mercury: mythology, science, and lessons for prevention". American Journal of Industrial Medicine. l (11): 779–787. doi:x.1002/ajim.20524. PMID 17918211.
- ^ a b c Couper, John (1837). "On the effects of black oxide of manganese when inhaled into the lungs". Br. Ann. Med. Pharm. Vital. Stat. Gen. Sci. i: 41–42.
- ^ Olsen, Sverre Eastward.; Tangstad, Merete; Lindstad, Tor (2007). "History of omanganese". Production of Manganese Ferroalloys. Tapir Academic Press. pp. 11–12. ISBN978-82-519-2191-6.
- ^ a b Preisler, Eberhard (1980). "Moderne Verfahren der Großchemie: Braunstein". Chemie in unserer Zeit (in German). 14 (5): 137–148. doi:ten.1002/ciuz.19800140502.
- ^ Bhattacharyya, P. K.; Dasgupta, Somnath; Fukuoka, Chiliad.; Roy Supriya (1984). "Geochemistry of braunite and associated phases in metamorphosed non-calcareous manganese ores of India". Contributions to Mineralogy and Petrology. 87 (1): 65–71. Bibcode:1984CoMP...87...65B. doi:ten.1007/BF00371403. S2CID 129495326.
- ^ a b c USGS Mineral Commodity Summaries 2009
- ^ Cook, Nigel J.; Ciobanu, Cristiana L.; Pring, Allan; Skinner, William; Shimizu, Masaaki; Danyushevsky, Leonid; Saini-Eidukat, Bernhardt; Melcher, Frank (2009). "Trace and pocket-sized elements in sphalerite: A LA-ICPMS report". Geochimica et Cosmochimica Acta. 73 (xvi): 4761–4791. Bibcode:2009GeCoA..73.4761C. doi:10.1016/j.gca.2009.05.045.
- ^ Wang, 10; Schröder, HC; Wiens, M; Schlossmacher, U; Müller, WEG (2009). "Manganese/polymetallic nodules: micro-structural characterization of exolithobiontic- and endolithobiontic microbial biofilms by scanning electron microscopy". Micron. forty (3): 350–358. doi:10.1016/j.micron.2008.10.005. PMID 19027306.
- ^ United Nations Ocean Economics and Technology Part, Technology Branch, United Nations (1978). Manganese Nodules: Dimensions and Perspectives. Marine Geology. Vol. 41. Springer. p. 343. Bibcode:1981MGeol..41..343C. doi:x.1016/0025-3227(81)90092-X. ISBN978-90-277-0500-6.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ "Manganese Mining in S Africa – Overview". MBendi.com. Archived from the original on 5 February 2016. Retrieved iv January 2014.
{{cite web}}: CS1 maint: bot: original URL condition unknown (link) - ^ Elliott, R; Coley, Thou; Mostaghel, S; Barati, M (2018). "Review of Manganese Processing for Production of TRIP/TWIP Steels, Part i: Current Do and Processing Fundamentals". JOM. lxx (five): 680–690. Bibcode:2018JOM...tmp...63E. doi:10.1007/s11837-018-2769-4. S2CID 139950857.
- ^ Corathers, 50. A.; Machamer, J. F. (2006). "Manganese". Industrial Minerals & Rocks: Commodities, Markets, and Uses (7th ed.). SME. pp. 631–636. ISBN978-0-87335-233-8.
- ^ a b Zhang, Wensheng; Cheng, Chu Yong (2007). "Manganese metallurgy review. Office I: Leaching of ores/secondary materials and recovery of electrolytic/chemic manganese dioxide". Hydrometallurgy. 89 (iii–iv): 137–159. doi:10.1016/j.hydromet.2007.08.010.
- ^ Grub, Norman; Nacu, Anca; Warkentin, Doug; Aksenov, Igor & Teh, Hoe (2010). "The Recovery of Manganese from low grade resources: bench scale metallurgical test program completed" (PDF). Kemetco Enquiry Inc. Archived from the original (PDF) on 2 February 2012.
- ^ "The CIA secret on the bounding main flooring". BBC News. 19 February 2018. Retrieved 3 May 2018.
- ^ "Project Azorian: The CIA'south Declassified History of the Glomar Explorer". National Security Annal at George Washington Academy. 12 Feb 2010. Retrieved eighteen September 2013.
- ^ Hein, James R. (January 2016). Encyclopedia of Marine Geosciences - Manganese Nodules. Springer. pp. 408–412. Retrieved 2 February 2021.
- ^ Hoseinpour, Vahid; Ghaemi, Nasser (1 December 2018). "Dark-green synthesis of manganese nanoparticles: Applications and future perspective–A review". Journal of Photochemistry and Photobiology B: Biological science. 189: 234–243. doi:10.1016/j.jphotobiol.2018.ten.022. PMID 30412855. S2CID 53248245. Retrieved two February 2021.
- ^ International Seabed Authority. "Polymetallic Nodules" (PDF). isa.org. International Seabed Authority. Retrieved 2 February 2021.
- ^ Oebius, Horst U; Becker, Hermann J; Rolinski, Susanne; Jankowski, Jacek A (January 2001). "Parametrization and evaluation of marine environmental impacts produced by deep-sea manganese nodule mining". Deep Sea Research Part Two: Topical Studies in Oceanography. 48 (17–eighteen): 3453–3467. Bibcode:2001DSRII..48.3453O. doi:ten.1016/s0967-0645(01)00052-two. ISSN 0967-0645.
- ^ Thompson, Kirsten F.; Miller, Kathryn A.; Currie, Duncan; Johnston, Paul; Santillo, David (2018). "Seabed Mining and Approaches to Governance of the Deep Seabed". Frontiers in Marine Science. 5. doi:10.3389/fmars.2018.00480. S2CID 54465407.
- ^ a b Ray, Durbar; Babu, Due east. V. S. S. K.; Surya Prakash, L. (1 January 2017). "Nature of Suspended Particles in Hydrothermal Plume at 3°40'Northward Carlsberg Ridge:A Comparison with Deep Oceanic Suspended Matter". Current Science. 112 (1): 139. doi:10.18520/cs/v112/i01/139-146. ISSN 0011-3891.
- ^ a b Hernroth, Bodil; Tassidis, Helena; Baden, Susanne P. (March 2020). "Immunosuppression of aquatic organisms exposed to elevated levels of manganese: From global to molecular perspective". Developmental & Comparative Immunology. 104: 103536. doi:x.1016/j.dci.2019.103536. ISSN 0145-305X. PMID 31705914. S2CID 207935992.
- ^ a b Sim, Nari; Orians, Kristin J. (October 2019). "Annual variability of dissolved manganese in Northeast Pacific forth Line-P: 2010–2013". Marine Chemistry. 216: 103702. doi:10.1016/j.marchem.2019.103702. ISSN 0304-4203. S2CID 203151735.
- ^ Bartlett, Richmond; Ross, Donald (2005). "Chemical science of Redox Processes in Soils". In Tabatabai, Chiliad.A.; Sparks, D.L. (eds.). Chemic Processes in Soils. SSSA Book Serial, no. viii. Madison, Wisconsin: Soil Science Society of America. pp. 461–487. LCCN 2005924447.
- ^ Dixon, Joe B.; White, G. Norman (2002). "Manganese Oxides". In Dixon, J.B.; Schulze, D.M. (eds.). Soil Mineralogy with Environmental Applications. SSSA Book Series no. 7. Madison, Wisconsin: Soil Scientific discipline Society of America. pp. 367–386. LCCN 2002100258.
- ^ Verhoeven, John D. (2007). Steel metallurgy for the non-metallurgist. Materials Park, Ohio: ASM International. pp. 56–57. ISBN978-0-87170-858-viii.
- ^ Manganese USGS 2006
- ^ Dastur, Y. N.; Leslie, West. C. (1981). "Mechanism of work hardening in Hadfield manganese steel". Metallurgical Transactions A. 12 (v): 749–759. Bibcode:1981MTA....12..749D. doi:ten.1007/BF02648339. S2CID 136550117.
- ^ Stansbie, John Henry (2007). Iron and Steel. Read Books. pp. 351–352. ISBN978-1-4086-2616-0.
- ^ Brady, George S.; Clauser, Henry R.; Vaccari. John A. (2002). Materials Handbook: an encyclopedia for managers, technical professionals, purchasing and production managers, technicians, and supervisors. New York, NY: McGraw-Hill. pp. 585–587. ISBN978-0-07-136076-0.
- ^ Tweedale, Geoffrey (1985). "Sir Robert Abbott Hadfield F.R.Southward. (1858–1940), and the Discovery of Manganese Steel Geoffrey Tweedale". Notes and Records of the Royal Guild of London. xl (i): 63–74. doi:10.1098/rsnr.1985.0004. JSTOR 531536.
- ^ "Chemical properties of 2024 aluminum allow". Metal Suppliers Online, LLC. Retrieved 30 April 2009.
- ^ a b Kaufman, John Gilbert (2000). "Applications for Aluminium Alloys and Tempers". Introduction to aluminum alloys and tempers. ASM International. pp. 93–94. ISBN978-0-87170-689-8.
- ^ Leigh A. Graham; Alison R. Fout; Karl R. Kuehne; Jennifer 50. White; Bhaskar Mookherji; Fred M. Marks; Glenn P. A. Yap; Lev Due north. Zakharov; Arnold L. Rheingold & Daniel Rabinovich (2005). "Manganese(I) poly(mercaptoimidazolyl)borate complexes: spectroscopic and structural characterization of MnH–B interactions in solution and in the solid state". Dalton Transactions (1): 171–180. doi:10.1039/b412280a. PMID 15605161.
- ^ Chen, Daquin; Zhou, Yang; Zhong, Jiasong (2016). "A review on Mn4+ activators in solids for warm white low-cal-emitting diodes". RSC Advances. half dozen (89): 86285–86296. Bibcode:2016RSCAd...686285C. doi:x.1039/C6RA19584A.
- ^ Baur, Florian; Jüstel, Thomas (2016). "Dependence of the optical properties of Mn4+ activated A2Ge4O9 (A=K,Rb) on temperature and chemical environs". Journal of Luminescence. 177: 354–360. Bibcode:2016JLum..177..354B. doi:10.1016/j.jlumin.2016.04.046.
- ^ Jansen, T.; Gorobez, J.; Kirm, M.; Brik, M. Yard.; Vielhauer, South.; Oja, 1000.; Khaidukov, Due north. M.; Makhov, V. Northward.; Jüstel, T. (ane Jan 2018). "Narrow Band Deep Cerise Photoluminescence of YtwoMg3Ge3O12:Mniv+,Li+ Inverse Garnet for High Ability Phosphor Converted LEDs". ECS Journal of Solid State Science and Technology. vii (1): R3086–R3092. doi:10.1149/2.0121801jss.
- ^ Jansen, Thomas; Baur, Florian; Jüstel, Thomas (2017). "Reddish emitting Chiliad2NbF7:Mn4+ and ChiliadtwoTaFvii:Mn4+ for warm-white LED applications". Journal of Luminescence. 192: 644–652. Bibcode:2017JLum..192..644J. doi:10.1016/j.jlumin.2017.07.061.
- ^ Zhou, Zhi; Zhou, Nan; Xia, Mao; Yokoyama, Meiso; Hintzen, H. T. (Bert) (six October 2016). "Research progress and application prospects of transition element Mn4+-activated luminescent materials". Journal of Materials Chemistry C. 4 (39): 9143–9161. doi:ten.1039/c6tc02496c.
- ^ "TriGain LED phosphor organization using crimson Mn4+-doped circuitous fluorides" (PDF). GE Global Research. Retrieved 28 Apr 2017.
- ^ a b Dell, R. K. (2000). "Batteries fifty years of materials development". Solid Land Ionics. 134 (1–2): 139–158. doi:10.1016/S0167-2738(00)00722-0.
- ^ Kuwahara, Raymond T.; Skinner III, Robert B.; Skinner Jr., Robert B. (2001). "Nickel coinage in the United States". Western Journal of Medicine. 175 (2): 112–114. doi:10.1136/ewjm.175.2.112. PMC1071501. PMID 11483555.
- ^ "Design of the Sacagawea dollar". Us Mint. Retrieved four May 2009.
- ^ Shepard, Anna Osler (1956). "Manganese and Fe–Manganese Paints". Ceramics for the Archaeologist. Carnegie Institution of Washington. pp. twoscore–42. ISBN978-0-87279-620-1.
- ^ Law, N.; Caudle, M.; Pecoraro, 5. (1998). Manganese Redox Enzymes and Model Systems: Properties, Structures, and Reactivity. Advances in Inorganic Chemistry. Vol. 46. p. 305. doi:x.1016/S0898-8838(08)60152-X. ISBN9780120236466.
- ^ Takeda, A. (2003). "Manganese action in brain function". Encephalon Inquiry Reviews. 41 (1): 79–87. doi:10.1016/S0165-0173(02)00234-5. PMID 12505649. S2CID 1922613.
- ^ Silva Avila, Daiana; Luiz Puntel, Robson; Aschner, Michael (2013). "Chapter 7. Manganese in Health and Illness". In Astrid Sigel; Helmut Sigel; Roland G. O. Sigel (eds.). Interrelations betwixt Essential Metal Ions and Human Diseases. Metal Ions in Life Sciences. Vol. 13. Springer. pp. 199–227. doi:x.1007/978-94-007-7500-8_7. ISBN978-94-007-7499-5. PMC6589086. PMID 24470093.
- ^ Hernroth, Bodil; Krång, Anna-Sara; Baden, Susanne (February 2015). "Bacteriostatic suppression in Norway lobster (Nephrops norvegicus) exposed to manganese or hypoxia nether pressure of ocean acidification". Aquatic Toxicology. 159: 217–224. doi:10.1016/j.aquatox.2014.11.025. ISSN 0166-445X. PMID 25553539.
- ^ a b Institute of Medicine (US) Console on Micronutrients (2001). "Manganese". Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Chromium, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Chromium. National Academy Press. pp. 394–419. ISBN978-0-309-07279-3. PMID 25057538.
- ^ Come across "Manganese". Micronutrient Data Middle. Oregon Land University Linus Pauling Institute. 23 April 2014.
- ^ "Overview on Dietary Reference Values for the EU population as derived by the EFSA Panel on Dietetic Products, Nutrition and Allergies" (PDF). 2017.
- ^ Tolerable Upper Intake Levels For Vitamins And Minerals (PDF), European Food Condom Authority, 2006
- ^ "Federal Register May 27, 2016 Nutrient Labeling: Revision of the Diet and Supplement Facts Labels. FR page 33982" (PDF).
- ^ "Daily Value Reference of the Dietary Supplement Label Database (DSLD)". Dietary Supplement Label Database (DSLD). Archived from the original on seven Apr 2020. Retrieved 16 May 2020.
- ^ Umena, Yasufumi; Kawakami, Keisuke; Shen, Jian-Ren; Kamiya, Nobuo (May 2011). "Crystal construction of oxygen-evolving photosystem II at a resolution of ane.9 Å" (PDF). Nature. 473 (7345): 55–60. Bibcode:2011Natur.473...55U. doi:x.1038/nature09913. PMID 21499260. S2CID 205224374.
- ^ Dismukes, G. Charles; Willigen, Rogier T. van (2006). "Manganese: The Oxygen-Evolving Circuitous & Models". Manganese: The Oxygen-Evolving Circuitous & Models Based in role on the article Manganese: Oxygen-Evolving Complex & Models by Lars-Erik Andréasson & Tore Vänngård which appeared in the Encyclopedia of Inorganic Chemistry, First Edition, Beginning Edition. Encyclopedia of Inorganic Chemistry. doi:ten.1002/0470862106.ia128. ISBN978-0470860786.
- ^ "Safety Data Sheet". Sigma-Aldrich. Retrieved 26 July 2021.
{{cite web}}: CS1 maint: url-status (link) - ^ Hasan, Heather (2008). Manganese. The Rosen Publishing Group. p. 31. ISBN978-one-4042-1408-8.
- ^ "Manganese Chemical Groundwork". Metcalf Institute for Marine and Ecology Reporting University of Rhode Island. April 2006. Archived from the original on 28 Baronial 2006. Retrieved 30 Apr 2008.
- ^ "Run a risk Assessment Information System Toxicity Summary for Manganese". Oak Ridge National Laboratory. Retrieved 23 April 2008.
- ^ Ong, Yard. L.; Tan, T. H.; Cheung, Due west. L. (1997). "Potassium permanganate poisoning – a rare crusade of fatal self poisoning". Emergency Medicine Journal. fourteen (1): 43–45. doi:10.1136/emj.xiv.1.43. PMC1342846. PMID 9023625.
- ^ Young, R.; Critchley, J. A.; Young, 1000. Thousand.; Freebairn, R. C.; Reynolds, A. P.; Lolin, Y. I. (1996). "Fatal acute hepatorenal failure post-obit potassium permanganate ingestion". Human & Experimental Toxicology. 15 (3): 259–61. doi:10.1177/096032719601500313. PMID 8839216. S2CID 8993404.
- ^ a b "Safe and Health Topics: Manganese Compounds (as Mn)". U.Due south. Occupational Safety and Health Assistants.
- ^ "NIOSH Pocket Guide to Chemic Hazards – Manganese compounds and smoke (as Mn)". Centers for Disease Control. Retrieved 19 November 2015.
- ^ Yin, Z.; Jiang, H.; Lee, E. S.; Ni, M.; Erikson, K. K.; Milatovic, D.; Bowman, A. B.; Aschner, M. (2010). "Ferroportin is a manganese-responsive protein that decreases manganese cytotoxicity and accumulation" (PDF). Journal of Neurochemistry. 112 (5): 1190–eight. doi:10.1111/j.1471-4159.2009.06534.10. PMC2819584. PMID 20002294.
- ^ a b Bouchard, M. F; Sauvé, S; Barbeau, B; Legrand, M; Bouffard, T; Limoges, E; Bellinger, D. C; Mergler, D (2011). "Intellectual impairment in schoolhouse-age children exposed to manganese from drinking water". Environmental Wellness Perspectives. 119 (one): 138–143. doi:10.1289/ehp.1002321. PMC3018493. PMID 20855239.
- ^ Barceloux, Donald; Barceloux, Donald (1999). "Manganese". Clinical Toxicology. 37 (2): 293–307. doi:10.1081/CLT-100102427. PMID 10382563.
- ^ Devenyi, A. Thou; Barron, T. F; Mamourian, A. C (1994). "Dystonia, hyperintense basal ganglia, and high whole blood manganese levels in Alagille's syndrome". Gastroenterology. 106 (iv): 1068–71. doi:ten.1016/0016-5085(94)90769-2. PMID 8143974.
- ^ Agency for Toxic Substances and Disease Registry (2012) 6. Potential for human being exposure, in Toxicological Profile for Manganese, Atlanta, GA: U.S. Department of Health and Homo Services.
- ^ Pourkhabbaz, A; Pourkhabbaz, H (2012). "Investigation of Toxic Metals in the Tobacco of Different Iranian Cigarette Brands and Related Health Issues". Iranian Periodical of Basic Medical Sciences. 15 (i): 636–644. PMC3586865. PMID 23493960.
- ^ Talhout, Reinskje; Schulz, Thomas; Florek, Ewa; Van Benthem, January; Wester, Piet; Opperhuizen, Antoon (2011). "Chancy Compounds in Tobacco Smoke". International Journal of Environmental Inquiry and Public Health. eight (12): 613–628. doi:10.3390/ijerph8020613. PMC3084482. PMID 21556207.
- ^ Bernhard, David; Rossmann, Andrea; Wick, Georg (2005). "Metals in cigarette smoke". IUBMB Life. 57 (12): 805–nine. doi:10.1080/15216540500459667. PMID 16393783. S2CID 35694266.
- ^ Baselt, R. (2008) Disposition of Toxic Drugs and Chemicals in Man, 8th edition, Biomedical Publications, Foster City, CA, pp. 883–886, ISBN 0-9626523-seven-vii.
- ^ Normandin, Louise; Hazell, A. Due south. (2002). "Manganese neurotoxicity: an update of pathophysiologic mechanisms". Metabolic Encephalon Disease. 17 (4): 375–87. doi:10.1023/A:1021970120965. PMID 12602514. S2CID 23679769.
- ^ a b Cersosimo, M. G.; Koller, W.C. (2007). "The diagnosis of manganese-induced parkinsonism". NeuroToxicology. 27 (three): 340–346. doi:10.1016/j.neuro.2005.10.006. PMID 16325915.
- ^ Lu, C. South.; Huang, C.C; Chu, Due north.S.; Calne, D.B. (1994). "Levodopa failure in chronic manganism". Neurology. 44 (9): 1600–1602. doi:10.1212/WNL.44.ix.1600. PMID 7936281. S2CID 38040913.
- ^ a b Guilarte TR, Gonzales KK (August 2015). "Manganese-Induced Parkinsonism Is Not Idiopathic Parkinson's Affliction: Environmental and Genetic Evidence". Toxicological Sciences (Review). 146 (2): 204–12. doi:10.1093/toxsci/kfv099. PMC4607750. PMID 26220508.
- ^ a b Kwakye GF, Paoliello MM, Mukhopadhyay S, Bowman AB, Aschner M (July 2015). "Manganese-Induced Parkinsonism and Parkinson'southward Illness: Shared and Distinguishable Features". Int J Environ Res Public Health (Review). 12 (7): 7519–twoscore. doi:ten.3390/ijerph120707519. PMC4515672. PMID 26154659.
- ^ Peres TV, Schettinger MR, Chen P, Carvalho F, Avila DS, Bowman AB, Aschner Yard (November 2016). "Manganese-induced neurotoxicity: a review of its behavioral consequences and neuroprotective strategies". BMC Pharmacology & Toxicology (Review). 17 (ane): 57. doi:10.1186/s40360-016-0099-0. PMC5097420. PMID 27814772.
- ^ Lazrishvili, I.; et al. (2016). "Manganese loading induces mouse-killing behaviour in nonaggressive rats". Periodical of Biological Physics and Chemistry. sixteen (iii): 137–141. doi:10.4024/31LA14L.jbpc.16.03.
- ^ "Drinking Water Contaminants". US EPA. Retrieved ii Feb 2015.
- ^ Prabhakaran, M.; Ghosh, D.; Chapman, One thousand.D.; Gunasekar, P.Thou. (2008). "Molecular mechanism of manganese exposure-induced dopaminergic toxicity". Encephalon Enquiry Bulletin. 76 (iv): 361–367. doi:10.1016/j.brainresbull.2008.03.004. ISSN 0361-9230. PMID 18502311. S2CID 206339744.
External links [edit]
- National Pollutant Inventory – Manganese and compounds Fact Canvass
- International Manganese Institute
- NIOSH Manganese Topic Page
- Manganese at The Periodic Table of Videos (University of Nottingham)
- All about Manganese Dendrites
Source: https://en.wikipedia.org/wiki/Manganese







0 Response to "Black Fire Clip Art Most Common Isotope of Chromium"
Postar um comentário